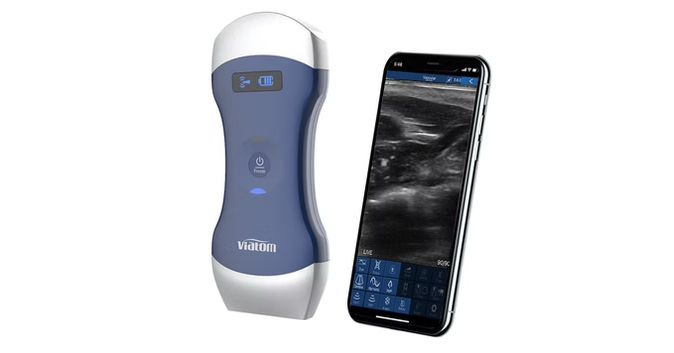
Butterfly iQ+
Handheld, whole-body ultrasound device with 20+ clinical presets and AI-enhanced imaging tools
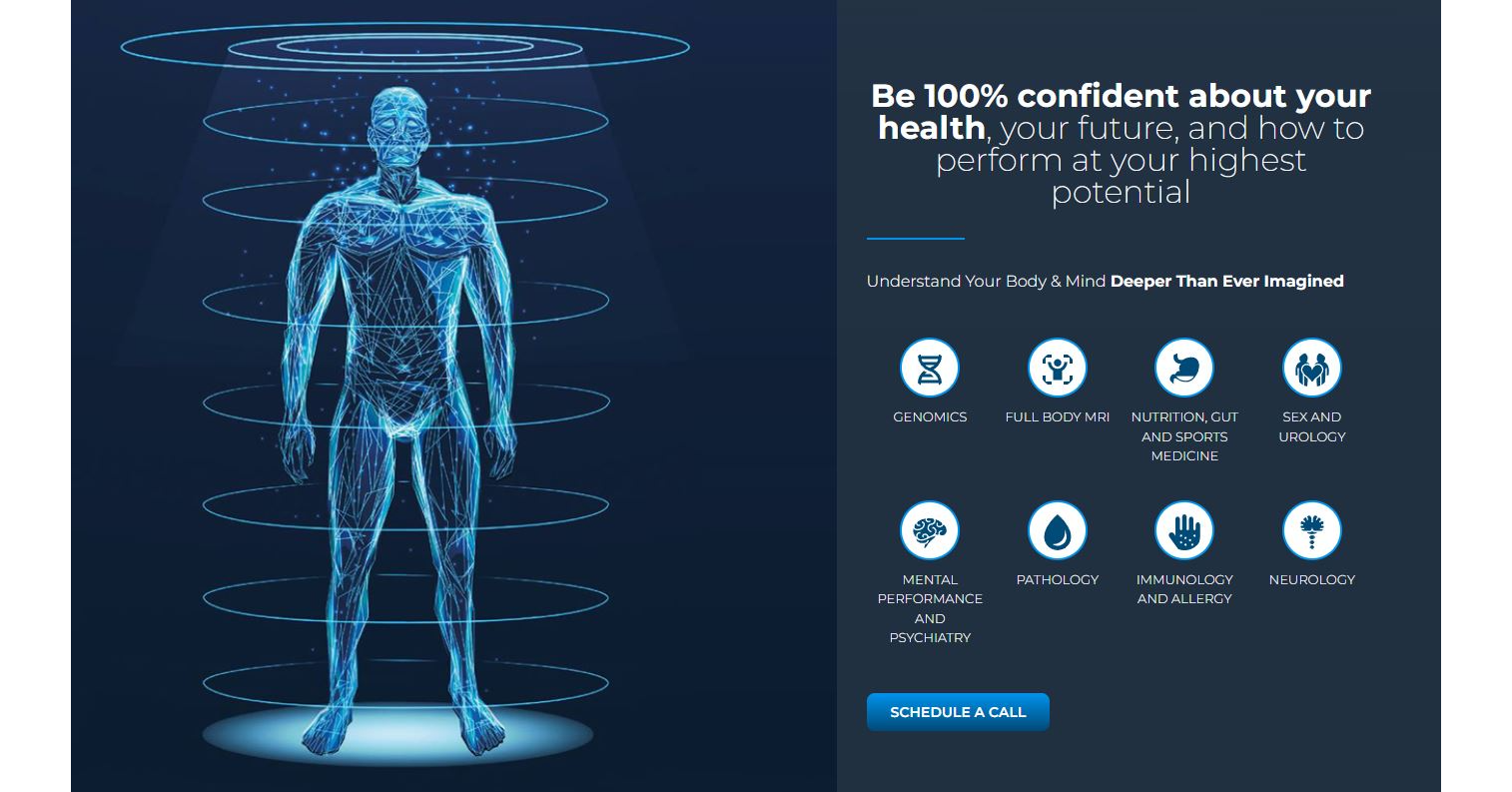
Butterfly iQ+ is a portable, single-probe whole-body point-of-care ultrasound system that connects to iOS or Android devices to enable diagnostic imaging across multiple clinical applications. Powered by Butterfly’s second-generation Ultrasound-on-Chip™ semiconductor technology, the device delivers high-performance imaging with optimized beamforming, fast frame rates, and a broad range of presets for abdominal, cardiac, lung, musculoskeletal, vascular, obstetric, and procedural guidance use cases. The system is designed for use by trained healthcare professionals in clinical environments and integrates with imaging workflows and data-management tools to support rapid assessment and measurement tasks.
Single handheld probe enabling whole-body ultrasound imaging.
Ultrasound-on-Chip™ technology for broad frequency range and wide clinical utility.
20+ clinical presets for abdominal, cardiac, lung, MSK, vascular, OB/GYN, and procedural imaging.
Auto bladder volume calculation and automated functional tools.
Needle Viz™ and advanced imaging aids for guided procedures.
Multiple scan modes including B-mode, M-mode, Color and Power Doppler.
Compatible with iOS and Android with secure cloud-enabled image storage.
Durable design with military-grade drop resistance and water/dust protection.
Integrated annotation, calipers, and workflow tools for clinical documentation.
- Butterfly reinvented ultrasound from the ground up, replacing piezoelectric crystal sensors with its postage stamp-sized Ultrasound-On-Chip™ technology—making medical imaging more accessible and affordable than ever before.
As the world’s first FDA-approved semiconductor chip-based, handheld, whole body ultrasound device, the ButterflyiQ+ solution far transcends what's possible with traditional ultrasound and has evolved well-beyond other point-of-care imaging products.
Specifically, Butterfly iQ+ is built with both custom-made and off-the-shelf components. The patents owned and in-licensed by Butterfly relate to the architecture of the company’s ultrasonic imaging devices, micro-fabricated ultrasonic transducers and machine learning for ultrasound applications. The key custom-made component in the Butterfly probe is the ultrasound transducer module consisting of a custom chip and lens. Creating the functional chip itself was the first major development breakthrough. Butterfly’s silicon chip offers 10,000 independent channels and 3D imaging, all while being small enough to fit on the 3x2 cm area at the end of the probe, and using low enough power to run on a battery.
The second milestone was developing the transducer sensor design, process and integration. The R&D team built the transducer directly on the electronics, then combined it with a custom lens and package, creating an entire ultrasound system on a single chip. Functionally, what makes Butterfly unique is its ability to close an information gap and facilitate better, more informed care at a time when caregivers need more connectedness to patients, better tools to know-more sooner, and interaction with technology that makes them better vs. distracts from what they love.
- Butterfly reinvented ultrasound from the ground up, replacing piezoelectric crystal sensors with its postage stamp-sized Ultrasound-On-Chip™ technology—making medical imaging more accessible and affordable than ever before.
- Laboratory & Diagnostics
- Released
- Clinical Reference
- Clinicians, Nurses
- Care Management
- Android, iOS
- Handheld, Mobile App
- FDA Cleared
- Obstetrics, Cardiology, Nursing, Physical Medicine and Rehabilitation, Emergency Medicine, Anesthesiology
- Device
- Hospital Inpatient, Hospital Outpatient, Physician Practice
- From September 2018 - June 2021, 4,695 pregnant volunteers were recruited in North Carolina and Zambia and obtained blind ultrasound sweeps (cineloop videos) of the gravid abdomen alongside standard fetal biometry. A UNC landmark study published in NEJM Evidence, found that, using Butterfly iQ and AI assistance, nurse midwives in Zambia with no prior sonography training can perform at the level of trained sonographers. A study done by South Metro Fire rescue in Colorado evaluated paramedics who were using Butterfly iQ+ devices to capture ultrasound video of patients in need of assistance to help with diagnosis. The paramedics recorded impressions of the ultrasound video for 49 patients and those videos were reviewed by a single POCUS expert investigator to determine adequacy of image quality and accuracy of interpretation. The study found that POCUS video images were considered adequate in 42/49 of patients, and they accurately interpreted the POCUS images in 43/49 of patients. That had a substantial impact on paramedic decision making and patient management in about 30% of patients. A pilot study of 44 subjects was conducted at a public hospital involving subjects with a history of heart failure. After a 15 min training session involving a tutorial video, subjects performed four-zone lung ultrasounds on themselves using a Butterfly device. This study found that Butterfly was effective when used by patients to assess the health of their lungs. This supports further investigation into a telemedicine model using lung ultrasounds to reduce emergency department visits and hospitalizations associated with heart failure.
Regulatory approvals for the Butterfly iQ+ ultrasound system
The Butterfly iQ® for iPhone received the US Food and Drug Administration (FDA) 510(k) clearance for human use in October 2017, becoming the world’s first approved ultrasound-on-a-chip-based imaging device.
Butterfly iQ® obtained CE Mark approval in April 2019, allowing licenced medical professionals in the UK, Europe, Australia and New Zealand to receive the ultrasound system. Health Canada cleared the device in March 2020.
In August 2021, Butterfly Network entered an exclusive strategic collaboration with Caption Health, an artificial intelligence (AI)-based ultrasound software developer, to incorporate a comprehensive AI-guided solution on the Butterfly iQ+ platform for early diagnosis and management of diseases.
The FDA approved Butterfly iQ3, a next-generation handheld point-of-care ultrasound system (POCUS), in January 2024. It is the third version of the world’s first semiconductor-based single-probe, whole-body ultrasound system Butterfly iQ.
- --
 My Rx Pad
My Rx Pad 


 Product Link
Product Link.png)

 FDA Cleared
FDA Cleared